Most patients with Cushing’s disease are successfully treated by transsphenoidal pituitary surgery and most cases of adrenal based Cushing’s syndrome require the removal of only one adrenal gland. In cases such as these, adrenal insufficiency (lack of sufficient hormone production from the adrenal glands) is almost always temporary and patients usually recuperate within one year.
However, there are cases where Cushing’s disease cannot be adequately treated with pituitary surgery or radiation and in other cases, adrenal based Cushing’s syndrome affects both glands. In patients such as these, bilateral adrenalectomy (BLA), resulting in permanent adrenal insufficiency, is often presented to patients as a treatment option. One of the first questions patients ask is does permanent adrenal insufficiency affect life expectancy and quality of life?
In short, we can say that the majority of patients do well with adequate replacement of adrenal hormones. However, there are potential threats in long term replacement which can decrease quality of life and shorten life expectancy in some patients.
Causes of adrenal failure
The number of patients that are permanently adrenal insufficient due to treatment of Cushing’s syndrome is actually quite small compared to other causes of permanent adrenal insufficiency. In some cases, a defect in pituitary or adrenal gland development during gestation results in lack of sufficient adrenal hormone production by the adrenal glands immediately at birth. One example of this is called congenital adrenal hyperplasia (CAH), which occurs in ~1/15000 births. Individuals with CAH lack an enzyme that is critical for cortisol production.
The most common cause of primary adrenal insufficiency (called Addison’s disease) appearing later in children or in adults is an auto-immune disease; an abnormal attack by the immune cells and antibodies resulting in destruction of the adrenal cortex, leading to lack of hormone production. Adrenal glands can also be destroyed by infections such as tuberculosis, inflammation, tumors or bleeding in both adrenals. There are also other types of adrenal disorders that result in bilateral adrenal tumors, such as bilateral pheochromocytomas which are adrenalin producing tumors.
In other patients, the lack of sufficient cortisol production is secondary to insufficient production of adrenocorticotropic hormone (ACTH) by the pituitary. ACTH is the key regulator of cortisol secretion by the adrenal glands. Secretion of ACTH can be permanently affected when extensive pituitary surgery is required to remove a large pituitary tumor. Most tumors causing Cushing’s disease are small, thus most Cushing’s disease patients retain sufficient ACTH production following surgery. However other types of pituitary tumors (non-functioning pituitary tumors, large prolactinomas, and acromegaly) can be much larger and these patients may experience permanent adrenal insufficiency. Several other causes of lack of ACTH production by the pituitary also exist. These include infections, auto-immune inflammation, radiotherapy and drugs such as narcotics.
Hormone replacement of adrenal failure
The lack of function of both adrenal glands can begin at birth or start at a very advanced age. In any patient with adrenal insufficiency, adequate replacement with appropriate medication is essential to maintain bodily function. The adrenal glands are composed of two main components as shown in Figure 1. The inner portion called the adrenal medulla produces adrenalin and noradrenalin. Patients facing a BLA will often ask if they will retain their normal fight and flight response without adrenal glands. Fortunately noradrenalin producing tissues are present throughout our body in the sympathetic and parasympathetic nervous system and adequately compensate for the lack of adrenal medulla hormones. Thus, normal fight/flight responses are retained and there is no need to replace the hormones produced by the adrenal medulla.
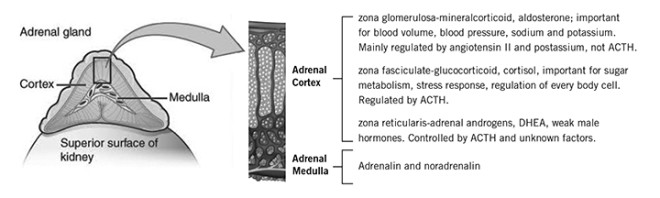
The outer portion of the adrenal gland is called the adrenal cortex and it produces cortisol, aldosterone and adrenal androgens (weak male hormones, particularly DHEAS or dehydroepiandrosterone sulfate). Cortisol, made in the zona fasciculata, is essential in regulating the proper function of almost every cell in our body and life is impossible without adequate intake of cortisol or equivalent synthetic glucocorticoid such as hydrocortisone, cortisone acetate, prednisone, or dexamethasone.
Daily production of cortisol is usually in the range of 15-25 mg per day. The highest production occurs just before awakening and in the morning hours with progressively less production in the afternoon and very little during the night. Replacement is usually provided as equivalent of hydrocortisone divided in three doses (very early upon awakening, 4-5 hours later and 4-6 hours later; for example 10 mg at 7:00 AM, 7.5 mg at 11AM and 5 mg at 3PM. Some patients do well with two doses daily, taking two thirds in morning and one third in afternoon.
Normally, in cases of infection, major stress, accident, or surgery, our pituitary and adrenal glands increase cortisol production 2-3 fold to defend against stress. Thus, it is mandatory that patients with adrenal insufficiency learn to increase 2-3 fold their glucocorticoid replacement during stress. If patients cannot retain their medication because of vomiting, they need to take cortisone replacement by intramuscular injection and go to the emergency room to receive intravenous replacement. This is necessary to avoid a life threatening adrenal crisis with low blood pressure, shock, low blood glucose, etc. All patients with adrenal insufficiency should carry a medical alert card and wear medical alert jewelry indicating their status in case they become confused or unable to provide a history of adrenal insufficiency.
Aldosterone, produced in the zona glomerulosa, is essential to maintain normal blood volume, sodium and potassium concentrations and adequate blood pressure. Aldosterone production is not mainly regulated by ACTH but rather by the renin-angiotensin system, thus production is usually preserved in patients with secondary pituitary causes, such as following pituitary surgery for Cushing’s disease or removal of a single adrenal gland. In other words, patients who require glucocorticoid replacement due to an ACTH deficiency generally do not require aldosterone replacement. If aldosterone is absent, for example in patients lacking adrenal glands, it needs to be replaced by the synthetic drug called fludrocortisone, in doses from 50-300 micrograms taken by mouth every morning. The dose is adjusted based on blood pressure, potassium and renin levels in blood. In case of acute stress/infection, only glucocorticoids, but not fludrocortisone needs to be increased.
Adrenal androgens, made in the zona reticularis usually do not need to be replaced in males because sufficient androgens are available from the testis. In females, adrenal androgen deficiency does not cause life threatening symptoms and the benefit of their replacement is assessed for each individual patient.
Threats in long-term replacement of adrenal failure
Adrenal crisis
The importance of proper education of patients and their family to increase replacement doses of glucocorticoids during acute infection or stress should not be underestimated. Recent surveys indicate that these concepts need to be repeated several times in order to maintain patients’ awareness. Despite providing information to patients and family, adrenal crisis episodes still occur in 3-7 patients out of 100 patients per year with adrenal failure either from adrenal or pituitary cause. Unfortunately, in some cases, this can be fatal. The most frequent causes are gastroenteritis and pulmonary infections (60–70%), and other events such as trauma, surgery, dental procedure, or major psychological distress. All patients and their close ones should be instructed how to inject intramuscularly supplementary hydrocortisone 100 mg (prescribing kit with syringe and needles) and to carry this with them when they travel particularly in remote areas where emergency rooms are not immediately accessible.
Steroid over-replacement
In the past, the normal amount of cortisol secretion was overestimated and led to patients being over-replaced with oral glucocorticoids. Excess cortisone replacement can lead to complications such as those found in patients with Cushing’s syndrome including weight gain, high blood pressure, increased glucose, cholesterol, osteoporosis, thinning of skin, infections, etc. With current glucocorticoid medications, it is difficult to mimic perfectly the normal fluctuations of cortisol secretion in humans. In other words, while on replacement medication, cortisol is too low when awakening in the morning and there are periods of cortisol excess following oral intake of glucocorticoid. Studies indicate that this can result in decreased sense of well-being in patients with adrenal insufficiency.
In addition, exposure to chronically increased levels of glucocorticoids can lead to an increased rate of diabetes, hypertension, high lipids, osteoporosis, doubling of cardiovascular risk and earlier death from heart attack, stroke, hip fracture etc. For this reason, current efforts are made to use adequate doses of replacement of glucocorticoid to avoid symptoms of adrenal insufficiency but also to avoid excess exposure to too much glucocorticoid. Important studies are currently under way to develop better medications to administer glucocorticoid replacement such that normal cortisol levels at different periods of the day are achieved and long periods of cortisol excess are avoided.
Conclusion
In order to maintain normal life expectancy and good quality of life, it is very important that patients with adrenal insufficiency and their close ones discuss these issues extensively with the medical team and nurses. Patients must learn to follow very closely the recommended daily timing and doses of hormone replacement, and to make proper adjustments in cases of illness. It is hoped that we will have even better preparations of steroid hormone replacement in the next decade to improve quality of life in patients with adrenal insufficiency.
By Dr. André Lacroix, Spring, 2017
Editor´s Note: Dr. André Lacroix is a Professor of Medicine, Centre at the hospitalier de l’Université de Montréal (CHUM).




Sorry, comments are closed for this post.